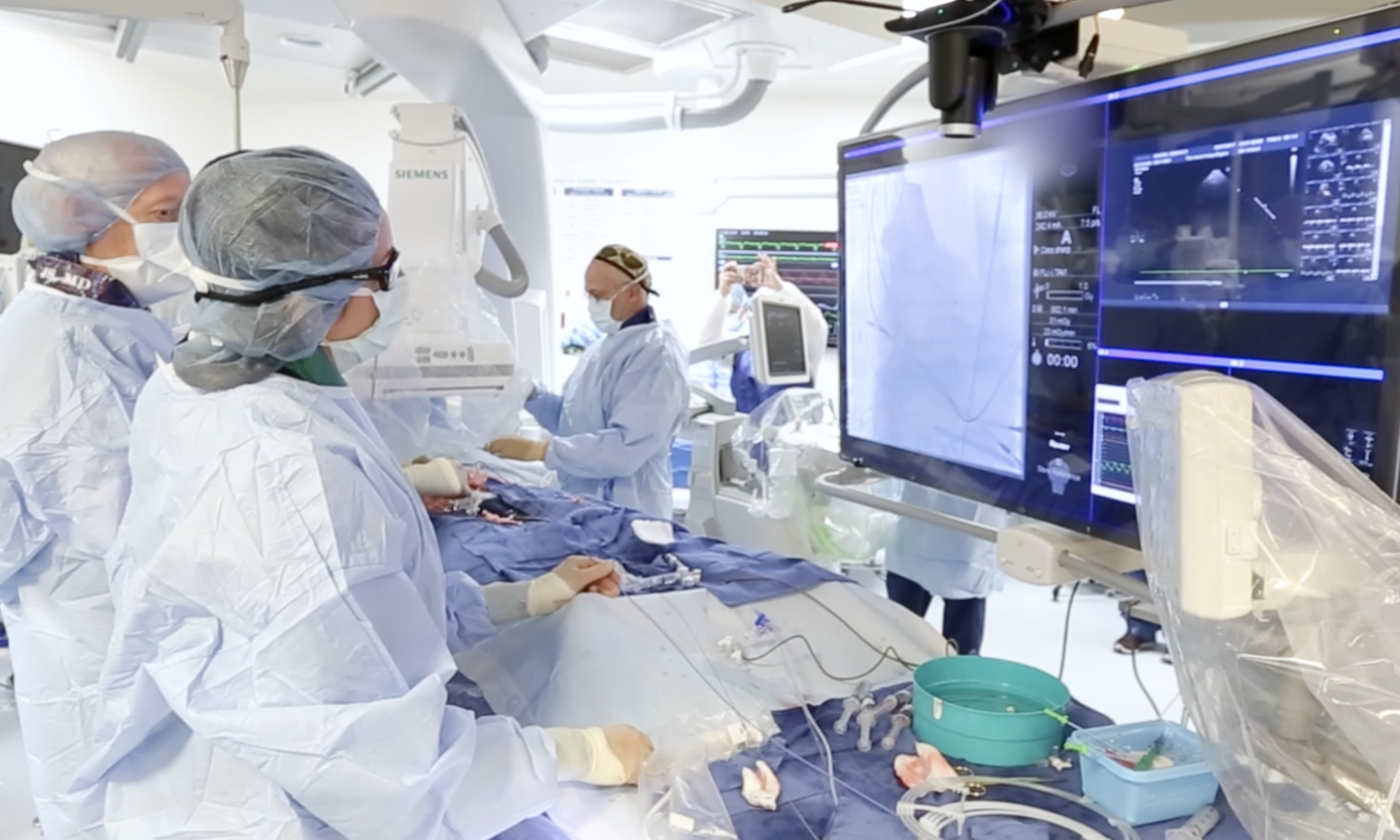
Two-year outcomes data on Transcatheter Aortic Valve Replacement (TAVR) in low-risk patients were recently presented at the American College of Cardiology 2020 Annual Scientific Session. PARTNER 3 demonstrated that TAVR with the Sapien 3 valve was as safe and effective as traditional open surgical replacement for low risk patients with aortic stenosis (AS). The primary outcome of the study—the risk of death, stroke or rehospitalization at two years—was comparable between the two groups.
Led by Michael J. Mack, MD, FACC — Chair of Cardiovascular Governance Council, Medical Director, Cardiovascular Surgery, Baylor Scott & White The Heart Hospital – Plano, and co-principal investigator of the PARTNER 3 study—Baylor Scott & White Health has been one of the top enrollers in this national study contributing to the overall findings over the past 2 years. Michael Mack received the Presidential Citation from American College of Cardiology (ACC) in 2012 and Masters of the ACC in 2020.